A major international study has found that the combination of two drugs - rivaroxaban and aspirin -- is superior to aspirin alone in preventing further heart complications in people with vascular disease.
The study of 27,400 people with stable coronary or peripheral artery disease from 33 countries worldwide will be published today, and results show that the combination of 2.5 mg of rivaroxaban twice daily plus 100 mg of aspirin once daily was significantly better than only aspirin or only rivaroxaban in preventing heart attacks, strokes, and death. Rivaroxaban, often known by the brand name Xarelto, is an anticoagulant, aspirin is an antiplatelet drug, and both are blood thinners.
 rivaroxaban
rivaroxaban 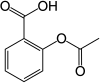 Aspirin
Aspirin
The results will be presented today at the Congress of the European Society of Cardiology (ESC) in Barcelona, Spain, and the overall results will be published in the New England Journal of Medicine.
The study, called COMPASS, is led by the Population Health Research Institute (PHRI) of McMaster University and Hamilton Health Sciences (HHS) in Hamilton, Canada. The study is funded by Bayer AG.
The findings are significant because there are about 300 million people around the world living with cardiovascular disease, and every year as many as five to 10 per cent have a stroke or heart attack. Although aspirin reduces the risk of major cardiovascular events by 19 per cent, a more effective antithrombotic strategy could have major benefits for the large population of patients with stable cardiovascular disease.
The clear result of this clinical study - that the combination reduced strokes, heart attacks and cardiovascular death by practically 25 per cent compared to either drug alone in both patients with stable coronary or peripheral artery disease - caused the clinical trial to be stopped early, after 23 months, in February 2017.
The researchers report that the drug combination does increase the chance of a major bleeding. These bleeds were mainly gastroenterological, and not in critical organs such as the brain nor fatal.
Co-principal investigator Dr. John Eikelboom and his team compared rivaroxaban at doses of 2.5 mg twice-daily combined with 100 mg of aspirin once-daily to rivaroxaban 5 mg twice-daily or to aspirin 100 mg once-daily. In the randomized clinical trial, patients were seen at one and six months, and then every six months.
They found the drug combination reduces cardiovascular outcomes, increases bleeding and improves survival in stable coronary or peripheral artery disease.
"Efforts to improve aspirin have focused primarily on combining aspirin with another antiplatelet drug or replacing aspirin with another antiplatelet drug, but this has had only limited success," said Eikelboom. He is a principal investigator of the PHRI, an associate professor of medicine at McMaster and a hematologist at HHS.


