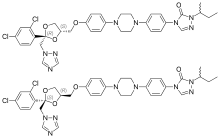
In continuation of my update on Itraconazole
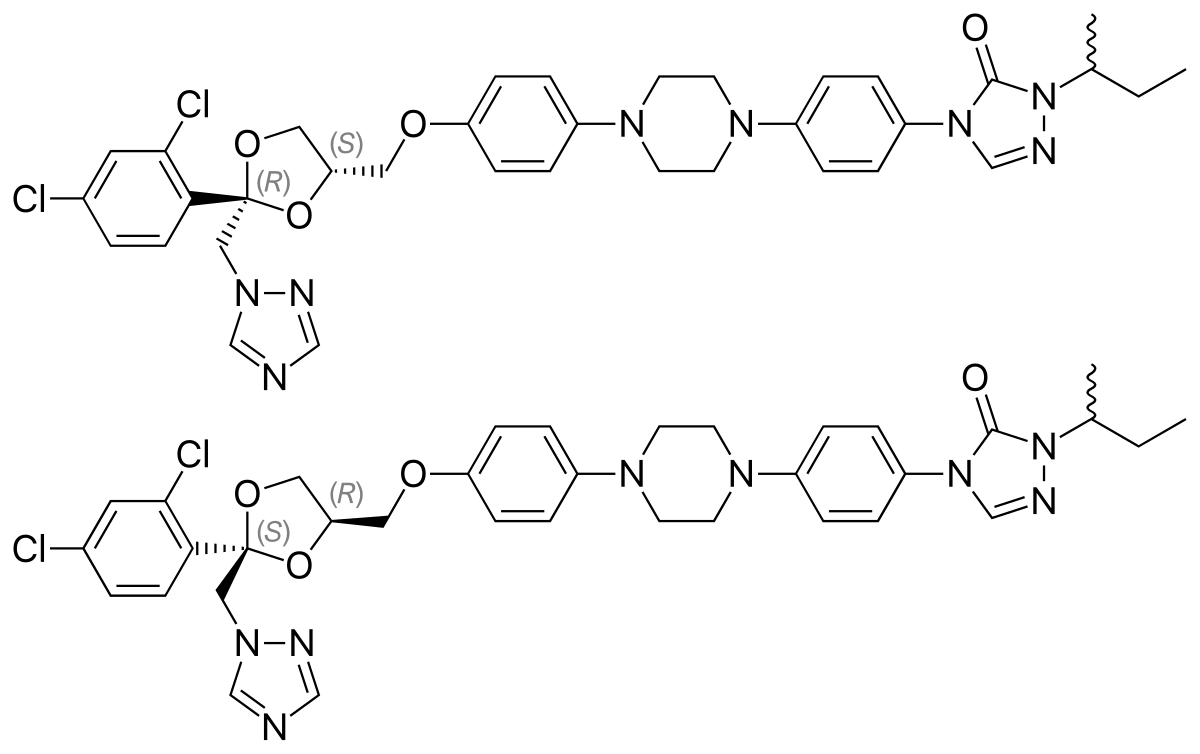
Mayne Pharma Group Limited (ASX: MYX) announced, that the US Food and Drug Administration (FDA) has approved the New Drug Application (NDA) for Tolsura™ (SUBA®-itraconazole) 65mg capsules. Tolsura is a new formulation of itraconazole indicated for the treatment of certain systemic fungal infections in adult patients.
Tolsura is indicated for the treatment of blastomycosis (pulmonary and extrapulmonary), histoplasmosis (including chronic cavitary pulmonary disease and disseminated, non-meningeal histoplasmosis) and aspergillosis (pulmonary and extrapulmonary, in patients who are intolerant of or who are refractory to amphotericin B therapy). These serious infections most commonly occur in vulnerable or immunocompromised patients, for example, those with a history of cancer, transplants (solid organ or bone marrow), HIV/AIDS, or chronic rheumatic disorders, and are often associated with high mortality rates or long-term health issues.
Mayne Pharma's CEO, Mr Scott Richards said, "We are very pleased to have received FDA approval of this patented formulation of itraconazole which incorporates Mayne Pharma's proprietary SUBA technology to improve the bioavailability of poorly soluble drugs. Reformulation of existing drugs plays an important role in improving patient compliance and clinical outcomes. We are proud to offer a new treatment option for patients with these life-threatening infections. We believe physicians will appreciate having access to Tolsura, which has been shown in clinical studies to have increased bioavailability and significantly reduced variability when compared to conventional oral itraconazole capsules."
"After many years of research and development and working closely with key global opinion leaders in infectious disease management, Tolsura represents a major milestone in the SUBA (SUper-BioAvailable) drug delivery platform at Mayne Pharma."
The Company will directly commercialise Tolsura and plans to launch in January 2019 with a new institutional sales team focused primarily on hospital-based infectious disease specialists. Tolsura has four granted patents from the United States Patent and Trademark Office with expiry dates ranging from 2023 to 2033.
The US anti-fungal triazole market has a current value of US$600m according to IQVIA and based on the clear unmet clinical need in serious systemic infections, the addressable market is estimated at US$200m[1].
Mayne Pharma directly markets more than 60 products in the US including three patent protected dermatology products Fabior® (tazarotene) foam, Sorilux® (calcipotriene) foam and Doryx® MPC (doxycycline) delayed-release tablets. The Company recently acquired Lexette® (halobetasol) foam used to treat plaque psoriasis which it expects to also launch in January 2019.
---------------------------------------------------------------------------------------------------------------------------