In continuation of my update on phenylephrine
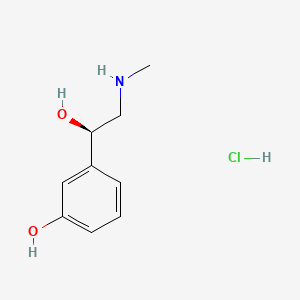
Eton Pharmaceuticals, Inc. (Nasdaq: ETON), a specialty pharmaceutical company focused on developing and commercializing innovative drug products, today announced that the U.S. Food and Drug Administration (FDA) has approved Biorphen, the first and only FDA-approved ready-to-use formulation of phenylephrine for the treatment of clinically important hypotension resulting primarily from vasodilation in the setting of anesthesia.
Prior to the FDA approval of Biorphen, phenylephrine injection was only approved and available as a highly concentrated formulation that required hospitals to manually dilute the concentrate prior to administration, or purchase ready-to-use formulations from 503B compounding pharmacies. Compounded drugs do not have to undergo FDA premarket review for safety, effectiveness and certain controls over manufacturing quality. Due to this lower regulatory standard, compounded drugs are often associated with higher risks of medication error.
“Today’s FDA approval of Biorphen addresses a critical medical need for an approved, ready-to-use standardized formulation of phenylephrine that can potentially reduce medication administration errors and improve patient safety,” said Sean Brynjelsen, chief executive officer of Eton Pharmaceuticals. "The approval of Biorphen represents an important step forward in Eton’s commitment to improving existing medicines to address unmet patient needs. We look forward to working with our manufacturing partner, Sintetica, to make Biorphen available to hospitals across the United States before the end of the year.”
“Operating rooms, emergency departments and intensive care units are fast-paced and time-sensitive environments. Add to that the complexity of caring for patients with a wide range of critical conditions and the use of high-alert medications like phenylephrine – and you have an atmosphere primed for potential medication error,” said Jared Marcucci, M.D., assistant director, Community First Medical Center Department of Emergency Medicine in Chicago. “As a practicing emergency medicine physician, the availability of an FDA-approved, ready-to-use formulation of phenylephrine is a welcome advance, providing physicians and hospital systems with an important new option that does not require compounding and can potentially help reduce the risk of medication errors and minimize harm to patients.”
“Providers at the bedside need reliable, easy to use, safe drugs that have a consistent supply. Many hospitals outsource unapproved ready-to-use phenylephrine from 503B compounders, however often have to navigate supply disruptions through these suppliers,” said Heather Nixon, M.D., Associate Professor Anesthesiology, University of Illinois at Chicago Hospital. “The availability of Biorphen will help address many of the underlying causes for risk and error associated with compounded phenylephrine while also reducing potential for waste associated with overdrawing medications. This will be an important new tool for anesthesiologists, pharmacists and other hospital providers in their efforts to enhance patient safety and prevent patient harm.”
FDA Approves Biorphen (phenylephrine hydrochloride) Ready-to-Use Injection for Hypotension During Anesthesia

1 comment:
I’m happy I located this blog! From time to time, students want to cognitive the keys of productive literary essays composing. Your first-class knowledge about this good post can become a proper basis for such people. nice one satta Matka
Post a Comment