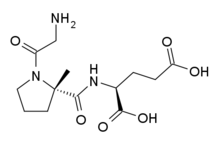
In continuation of my update on trofinetide
Acadia Pharmaceuticals Inc. (Nasdaq: ACAD) announced the U.S. Food and Drug Administration (FDA) approval of Daybue (trofinetide) for the treatment of Rett syndrome in adult and pediatric patients two years of age and older. Daybue is the first and only drug approved for the treatment of Rett syndrome.
“Today marks an important milestone for the Rett community and Acadia. As the first FDA-approved drug for the treatment of Rett syndrome, Daybue now offers the potential to make meaningful differences in the lives of patients and their families who have lacked options to treat the diverse and debilitating array of symptoms caused by Rett syndrome,” said Steve Davis, Acadia’s Chief Executive Officer. “We are grateful to all of the Rett syndrome patients, caregivers, clinical investigators and our employees who have contributed to making today a reality and look forward to getting Daybue to patients as quickly as possible.”
“Rett syndrome is a profoundly debilitating and complex, rare, neurodevelopmental disorder that presents differently across patients and can lead to an array of unpredictable symptoms throughout the course of a patient’s life,” said Jeffrey L. Neul, M.D., Ph.D., Annette Schaffer Eskind Chair and Director, Vanderbilt Kennedy Center, Professor of Pediatrics, Division of Neurology, Pharmacology, and Special Education, Vanderbilt University Medical Center and Phase 3 LAVENDER™ study investigator. “Now, for the first time after decades of clinical research, healthcare providers finally have a treatment option to address a range of core behavioral, communication and physical symptoms for their patients living with Rett syndrome.”
Rett syndrome is a complex, rare, neurodevelopmental disorder typically caused by a genetic mutation on the MECP2 gene.2,3 It is characterized by a period of normal development until six to 18 months of age, followed by significant developmental regression with loss of acquired communication skills and purposeful hand use. Symptoms of Rett syndrome may also include development of hand stereotypies, such as hand wringing and clapping, and gait abnormalities. Rett syndrome is believed to affect 6,000 to 9,000 patients in the U.S., with a diagnosed population of approximately 4,500 U.S. patients.
The FDA approval of Daybue was supported by results from the pivotal Phase 3 LAVENDER study evaluating the efficacy and safety of trofinetide versus placebo in 187 female patients with Rett syndrome five to 20 years of age. In the study, treatment with Daybue demonstrated statistically significant improvement compared to placebo on both co-primary efficacy endpoints, as measured by the change from baseline in Rett Syndrome Behaviour Questionnaire (RSBQ) total score (p=0.018) and the Clinical Global Impression-Improvement (CGI-I) scale score (p=0.003) at week 12. The RSBQ is a caregiver assessment that evaluates a range of symptoms of Rett syndrome including vocalizations, facial expressions, eye gaze, hand movements (or stereotypies), repetitive behaviors, breathing, night-time behaviors and mood. The CGI-I is a global physician assessment of whether a patient has improved or worsened. In the study, the most common side effects were diarrhea (82%) and vomiting (29%).
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” said Melissa Kennedy, Chief Executive Officer of the International Rett Syndrome Foundation. “Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones.”
FDA Approves Daybue (trofinetide) for the Treatment of Rett Syndrome