The FDA approval is based on results from a clinical program that evaluated the safety and efficacy of Brenzavvy in 23 clinical trials enrolling more than 5,000 adults with type 2 diabetes mellitus. Phase 3 studies showed Brenzavvy significantly reduced hemoglobin A1c and fasting blood sugar after 24 weeks, either as a monotherapy, in combination with metformin, or as an add-on to standard-of-care treatment consisting of a variety of regimens, including metformin, sulfonylureas, insulin, DPP4 inhibitors, or combinations of these agents. Although Brenzavvy is not approved for weight or blood pressure reduction, modest decreases in both weight and systolic blood pressure have been observed in the clinical program.
“As a class of drugs, SGLT2 inhibitors have shown tremendous benefit in treating adults with type 2 diabetes,” said Dr. Mason Freeman, M.D., Director of the Translational Research Center at Massachusetts General Hospital. “Being involved in all of the clinical trials for Brenzavvy, I am greatly impressed with the efficacy of the drug in reducing blood glucose levels and I believe it is an important addition to the SGLT2 inhibitor class of drugs.”
Brenzavvy treatment can be initiated in adults with type 2 diabetes with an estimated glomerular filtration rate (eGFR) greater than 30 mL/min/1.73 m2. Patients with eGFR less than 60 and greater than 30 mL/min/1.73 m2 are said to be in stage 3 chronic kidney disease, and for these patients metformin is often avoided due to the risk of lactic acidosis.
“Today's FDA approval represents a significant milestone for TheracosBio and provides an important treatment option to patients who suffer from type 2 diabetes. We look forward to bringing Brenzavvy to market,” said Albert R. Collinson, Ph.D., President and CEO of TheracosBio. “The approval of the Brenzavvy NDA is a result of the tireless work of the TheracosBio team and investigators. I want to thank all of the patients who took part in our clinical trials.”
According to the U.S. Centers for Disease Control and Prevention, more than 33 million Americans have type 2 diabetes, which means their bodies don’t use insulin correctly and as a result their blood sugar levels are too high. While some people can control their blood sugar levels with exercise and a healthy diet, others may need additional help to achieve good blood sugar (glycemic) control.
SGLT2 inhibitors are a class of prescription medicines that lower blood sugar by causing the kidneys to remove sugar from the body through urine.
Brenzavvy is available as 20 mg oral tablets recommended to be taken once daily, in the morning with or without food.

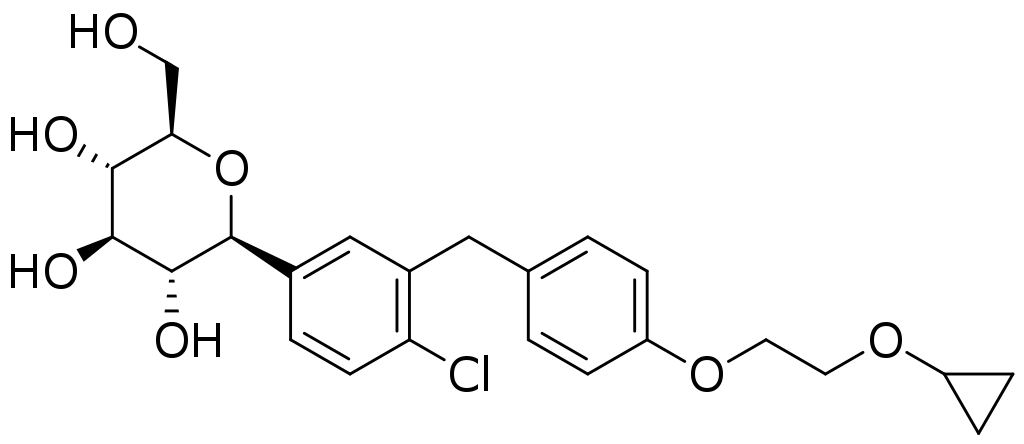


 Saxagliptin and
Saxagliptin and 





