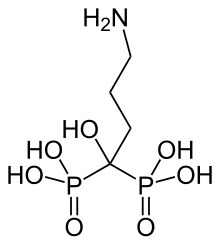
EffRx Pharmaceuticals SA, an Epalinges/Lausanne, Switzerland based drug delivery company announces that the New Drug Application (NDA) for the company's lead development program EX101 has been accepted for filing by the US Food and Drug Administration. EX101 is a proprietary buffered effervescent dosage form of alendronate sodium administered once weekly for treatment of osteoporosis in postmenopausal women and to increase bone mass in men with osteoporosis. The EX101 formulation is the first and only effervescent bisphosphonate alternative to tablets. EX101 has a pleasant taste of strawberry and is quickly and completely dissolved.
About Alendronate : Alendronic acid or alendronate sodium ( sold as Fosamax by Merck) is a bisphosphonate drug used for osteoporosis and several other bone diseases. It is marketed alone as well as in combination with vitamin D (2,800 U and 5600 U, under the name Fosamax+D). Merck's U.S. patent on alendronate expired in 2008 and Merck lost a series of appeals to block a generic version of the drug from being certified by the FDA. On February 6, 2008, the US FDA approved the first generic versions of alendronate, which were marketed by Barr Pharmaceuticals and Teva Pharmaceuticals USA. Teva Pharmaceuticals manufactures generic alendronate in 5-milligram, 10-milligram, and 40-milligram daily doses, and in 35-milligram and 70-milligram weekly doses, while Barr made generic alendronate in 70-milligram tablets, which were taken once weekly. Barr pharmaceuticals were subsequently acquired by Teva in July 2008...
Ref : http://www.effrx.com/firsteffrxnda.htm