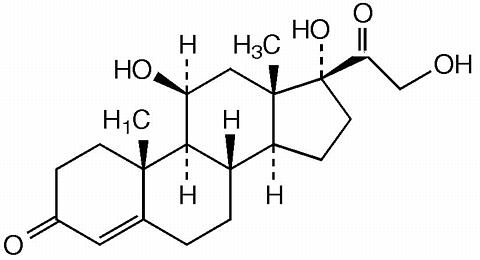
In continuation of my update on hydrocortisone
Eton Pharmaceuticals, Inc (Nasdaq: ETON), a specialty pharmaceutical company focused on developing and commercialising innovative treatments for rare pediatric diseases, announced the U.S. Food and Drug Administration (FDA) approval of Alkindi Sprinkle (hydrocortisone) oral granules as replacement therapy for Adrenocortical Insufficiency (AI) in children under 17 years of age. Alkindi Sprinkle is the first and only FDA-approved granular hydrocortisone formulation for the treatment of adrenocortical insufficiency specifically designed for use in children.
“The FDA approval of Alkindi Sprinkle is a breakthrough for patients and caregivers treating pediatric adrenocortical insufficiency. We are excited to offer an FDA-approved product that enables low dosing and administration of hydrocortisone to pediatric patients,” said Sean Brynjelsen, CEO of Eton Pharmaceuticals. “We look forward to making the product available to patients in the coming months.”
“For years, we heard from parents about their struggle to provide the right dose to their child,” said Dina M. Matos, Executive Director of the CARES Foundation. “We are thrilled the FDA has approved Alkindi Sprinkle for pediatric patients with Adrenocortical Insufficiency including patients with Congenital Adrenal Hyperplasia, a type of Adrenocortical Insufficiency.”
The FDA approval of Alkindi Sprinkle was supported by six clinical studies, including the first and only interventional Phase III study of oral hydrocortisone for Pediatric AI in neonates to children under eight years of age. Prior to the approval of Alkindi Sprinkle, oral hydrocortisone was only FDA-approved in tablet formulations of 5mg and stronger. Many pediatric patients require significantly lower doses and the flexibility of precision titration. Alkindi Sprinkle will be available in 0.5mg, 1mg, 2mg, and 5mg strengths, allowing clinicians greater flexibility to individualize dosing based on each patient’s needs in accordance with the instructions for dosage and administration.
Eton expects Alkindi Sprinkle to be commercially available in the fourth quarter of 2020.