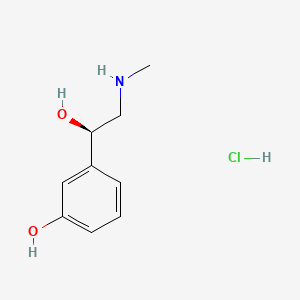
In continuation of my update on phenylephrine hydrochloride
Eyenovia, Inc, announced the U.S. Food and Drug Administration (FDA) approval of Mydcombi (tropicamide and phenylephrine hydrochloride ophthalmic spray) 1%/2.5% for inducing mydriasis for diagnostic procedures and in conditions where short term pupil dilation is desired. This represents the first approved fixed dose combination of tropicamide and phenylephrine in the United States and also the first product using Eyenovia’s proprietary Optejet device to be approved by any regulatory authority.
Mydcombi is designed to
improve the efficiency of the estimated 106 million office-based comprehensive
eye exams performed every year in the United States, as well as the estimated 4
million pharmacologic mydriasis applications for cataract surgery. The product
is contraindicated and should not be used in patients with known
hypersensitivity to any component of the formulation.
“The approval of Mydcombi, our first FDA approved product,
represents the culmination of years of tireless effort by the entire Eyenovia
team, and I would like to express my sincere gratitude to the associates and
technical experts who helped advance this important program through this
transformational milestone,” stated Michael Rowe, chief executive officer of
Eyenovia. “We look forward to introducing Mydcombi to key offices beginning
this summer while we bring our internal manufacturing capabilities on-line for
2024.”
“Perhaps more importantly, FDA approval of Mydcombi provides
critical validation of the Optejet as it is the first product approved using
the Optejet platform, which is core not only to our internal development
programs, including MicroLine for presbyopia, but our partnered programs as
well. We see opportunities to unlock significant opportunities in the future
treatment of other ophthalmic conditions including glaucoma and dry eye. I am
confident in our ability to maintain our current momentum.”
“I am proud of our team for this significant achievement – which
represents many ‘firsts’ for eye care,” stated Dr. Sean Ianchulev, Founder and
Chairman of Eyenovia’s Board of Directors. “The use of eye dropper bottles has
presented challenges for dosing in ophthalmologic settings in millions of
patients. We can do better now using sophisticated micro-array print delivery
with physiologic dosing that is similar to the natural tear film volume.”
https://en.wikipedia.org/wiki/Phenylephrine
https://en.wikipedia.org/wiki/Tropicamide