In continuation of my update on Paclitaxel
Results from the I-SPY 2 trial show that giving patients with HER2-positive invasive breast cancer a combination of the drugs trastuzumab emtansine (T-DM1) and pertuzumab before surgery was more beneficial than the combination of paclitaxel plus trastuzumab. Previous studies have shown that a combination of T-DM1 and pertuzumab is safe and effective against advanced, metastatic HER2-positive breast cancer, but in the new results, investigators tested whether the combination would also be effective if given earlier in the course of treatment. Results of the study are presented by trial investigators from the Abramson Cancer Center at the University of Pennsylvania at the AACR Annual Meeting 2016, April 16-20.
In this latest phase of the I-SPY2 trial, investigators worked to determine whether T-DM1 plus pertuzumab could eradicate residual disease (known as pathological complete response, or pCR) for more patients if delivered before surgery to shrink cancer tumors compared with paclitaxel plus trastuzumab. They also examined whether this combination could meet that goal without the need for patients to receive paclitaxel.
"The combination of T-DM1 and pertuzumab substantially reduced the amount of residual disease in the breast tissue and lymph nodes for all subgroups of HER2-positive breast cancers compared with those in the control group," said lead author, Angela DeMichele, MD, MSCE, a professor of Medicine and Epidemiology at the Perelman School of Medicine at the University of Pennsylvania, who will present the findings. "Our results suggest a possible new treatment option for patients that can not only effectively shrink tumors in the breast, but potentially reduce the chance of the cancer coming back later. The results also show that by replacing older, non-targeted therapies with more effective and less-toxic new therapies, we have the potential to both improve outcomes and decrease side effects."
For the study, patients whose tumors were 2.5 cm or bigger were randomly assigned to 12 weekly cycles of paclitaxel plus trastuzumab (control) or T-DM1 plus pertuzumab (test). Following the initial test period, all patients received four cycles of the chemotherapies doxorubicin and cyclophosphamide, and surgery. Patients' tumors were then tested for one of three biomarker signatures: HER2-positive, HER2-positive and hormone receptor (HR)-positive, and HER2-positive and HR-negative.
New drug combination before surgery may improve outcomes in breast cancer patients: Results from the I-SPY 2 trial show that giving patients with HER2-positive invasive breast cancer a combination of the drugs trastuzumab emtansine (T-DM1) and pertuzumab before surgery was more beneficial than the combination of paclitaxel plus trastuzumab.

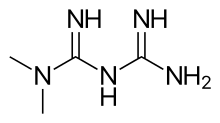
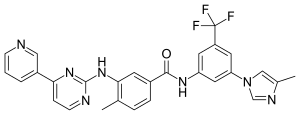

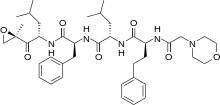 Carfilzomib
Carfilzomib 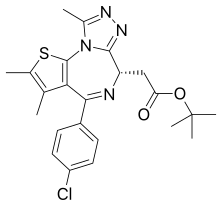 JQ1
JQ1  ABT-199
ABT-199

 Sofosbuvir
Sofosbuvir