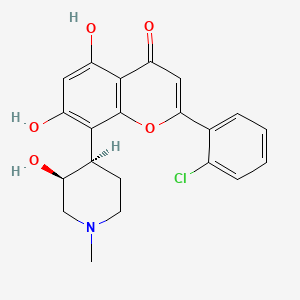
Top, Venn diagram of substructures in compounds that were found to bind to RNA from the fluorescence screening assay showed in Fig. 1c. Data were compiled by using compounds that had a P value of <0.001 for binding to the RNA hairpins. Bottom, structures of compounds 1 and 2 that were the most avid for binding to AUAU and AAUU RNA hairpins.
In an important new study with implications for the treatment of dozens of incurable diseases, scientists from the Florida campus of The Scripps Research Institute (TSRI) have for the first time created a drug candidate that attacks and neutralizes the RNA structure that causes an incurable progressive, inherited disease involving a gradual loss of control over body movement.
The study, which was published June 1, 2016 in Nature Communications, showed the compound significantly improved several aspects of cells taken directly from patients with spinocerebellar ataxia type 10 (SCA10), a form of spinocerebellar ataxia.
“More than 30 diseases, all of them incurable, are caused by RNA repeats,” said TSRI Professor Matthew Disney, who led the study. “By a thorough basic science investigation, we identified small molecules that target RNA base pairs precisely. We then leveraged this information to design the first drug candidate that binds to disease-causing defects in SCA10. Application of the drug candidate returns certain aspects of those cells to healthy levels—it’s like the defect is not even there.”
SCA10 is caused by what is called a pentanucleotide repeat (a genetic sequence of five nucleotides repeated many more times than normal) affecting the mitochondria, the cell’s energy source. The new drug candidate, known as 2AU-2, targets these repeats by binding to RNA base pairs.
“The potent bioactivity of 2AU-2 to moderate the structurally induced toxicity in SCA10 strongly suggests that base-pair-targeting RNA modules could have broad applicability in our effort to develop other compounds that target different RNAs,” said TSRI Research Associate Wang-Yong Yang, the first author of the study. “More than 70 percent of RNA secondary structure is made up of base pairing.”
The Disney group has developed new tools to identify optimal interactions between RNA structures and drug candidates targeting them. A database of these interactions has already been used to design several small molecule drug candidates.
“We are in the process of developing tools that allow one to design small molecules to target any RNA structural motif in a complex cellular environment. That environment can contain millions of other RNAs. In this study, Wang-Yong has done an exceptional job tackling this previously-thought-to-be-impossible molecular recognition problem,” Disney said.
Pathogenic RNA repeats contribute to disorders including Huntington’s disease, fragile X-associated tremor ataxia syndrome and myotonic dystrophy type 1 and 2.
Ref : http://www.nature.com/ncomms/2016/160601/ncomms11647/full/ncomms11647.html
Ref : http://www.nature.com/ncomms/2016/160601/ncomms11647/full/ncomms11647.html





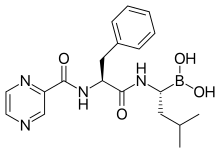
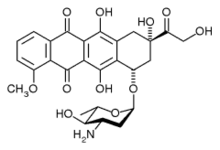 epirubicin
epirubicin letrozole
letrozole 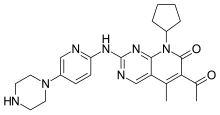 palbociclib
palbociclib