In continuation of my update on Pycnogenol....
A new peer-reviewed, published study shows French maritime pine bark extract, Pycnogenol®, may be effective in curbing muscle loss that occurs with aging – a natural process that leads to sarcopenia, a common condition affecting adults as early as age 65. The study, published in the September 2016 Journal on Orthopedics and Traumatology, revealed that daily supplementation with Pycnogenol® helps to stabilize muscles through this natural process and supports muscular function and endurance.

According to the National Library of Medicine, physically inactive individuals can lose as much as eight percent of muscle mass each decade after 30. Those who are physically active can maintain more muscle mass yet are still affected by muscle loss due to aging.
"Muscle loss due to aging can affect even the most active individuals and can result in feelings of weakness and frustrating fatigue which can interfere with our daily activities. This natural process is why opening a jar of pickles gets harder as we get older. We have less muscle to work with, so our muscles must work harder and, thus, we get tired faster," said nationally-known natural nutrition expert Dr. Fred Pescatore.
"This new study builds on a body of research connecting Pycnogenol® to healthy aging," said Dr. Pescatore. "As we age, muscle loss starts to impact the daily activities we tend to take for granted like climbing stairs, lifting groceries or enjoying walks without getting tired. This research shows that Pycnogenol can help alleviate the symptoms of sarcopenia and can support muscle function as we age."
In the study, participants between ages 70 - 78 and exhibiting symptoms of muscle loss, sarcopenia and fatigue but otherwise healthy, reported feeling increased muscle endurance in completing daily tasks such as walking and stair climbing, and improved overall muscular function by more than 40 percent. After just 8 weeks of taking 150 mg of Pycnogenol® the study results showed:
"What I found really compelling is the measurable effect Pycnogenol® had on the participants in relation to completing daily tasks -- the research shows that Pycnogenol® can play a role in helping you stay active with greater muscular stability and maintain muscle function as you age," Dr. Pescatore noted.
While experts recommend adequate amounts of sleep, in addition to diet and daily exercise to maintain physical fitness in later years, the study results are encouraging for those looking for an added boost in muscle function overall.
Ref : http://www.webmd.com/vitamins-supplements/ingredientmono-1019-pycnogenol.aspx?activeingredientid=1019

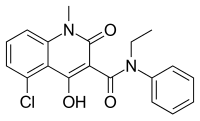

 Midostaurin
Midostaurin 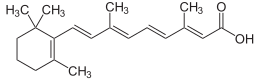


 Topiramate
Topiramate 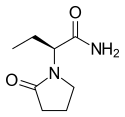 Levetiracetam
Levetiracetam